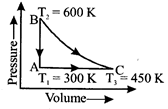
A) \[{{q}_{AB}}=450R\] and \[{{q}_{CA}}=-450R\]
B) \[{{q}_{AB}}=450R\] and \[{{q}_{CA}}=-225R\]
C) \[{{q}_{AB}}=450R\] and \[{{q}_{CA}}=-375R\]
D) \[{{q}_{AB}}=375R\] and \[{{q}_{CA}}=-450R\]
Correct Answer: C
Solution :
For monoatomic gas \[{{C}_{V}}=\frac{3}{2}R\,\,{{C}_{p}}=\frac{5}{2}R\] At constant volume, \[\Delta U={{q}_{V}}=n{{C}_{v,m}}\,\Delta T\] At constant pressure, \[\Delta H={{q}_{p}}=n{{C}_{p,m}}\Delta T\]You need to login to perform this action.
You will be redirected in
3 sec
