A) \[102\text{ }kcal\text{ }mo{{l}^{-1}}\]
B) \[51\text{ }kcal\text{ }mo{{l}^{-1}}\]
C) \[26\text{ }kcal\text{ }mo{{l}^{-1}}\]
D) \[204\text{ }kcal\text{ }mo{{l}^{-1}}\]
Correct Answer: B
Solution :
[b] Bond dissociation energy of \[P{{H}_{3}}\left( g \right)=228\text{ }kcal\text{ }mo{{l}^{-1}}\] \[P-H\] bond energy\[=\frac{228}{3}=76\text{ }kcal\text{ }mo{{l}^{-1}}\]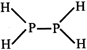 Bond energy of \[4(P-H)+(P-P)\] \[=355\text{ }kcal\text{ }mo{{l}^{-1}}\] or \[4\times 76+\left( P-P \right)=355\text{ }kcal\text{ }mo{{l}^{-1}}\] \[=P-P\] bond energy \[=51\text{ }kcal\text{ }mo{{l}^{-1}}\]
Bond energy of \[4(P-H)+(P-P)\] \[=355\text{ }kcal\text{ }mo{{l}^{-1}}\] or \[4\times 76+\left( P-P \right)=355\text{ }kcal\text{ }mo{{l}^{-1}}\] \[=P-P\] bond energy \[=51\text{ }kcal\text{ }mo{{l}^{-1}}\]
You need to login to perform this action.
You will be redirected in
3 sec
