| Electron distribution of two elements P and Q in their outermost shell is shown below. |
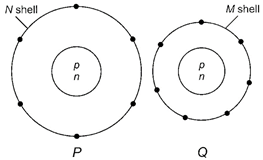
|
| Atomic numbers of P and Q are respectively |
A) 9, 17
B) 15, 17
C) 24, 17
D) 17, 24
Correct Answer: C
Solution :
Electron configuration of P :| K | L | M | N |
| 2 | 8 | 8 | 6 |
| So, its atomic number = 24 |
| Electronic configuration of Q : |
| K | L | M | N |
| 2 | 8 | 7 |
You need to login to perform this action.
You will be redirected in
3 sec
