| Consider following sets: |
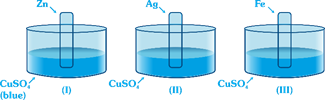 |
| Blue colour solution changes to colourless (or fades) in |
A) I, II, III
B) I, II
C) II, III
D) I, III
Correct Answer: D
Solution :
| [d] Since Zn, Fe is above in Electrochemical series (better reducing agent) thus it replaces Cu from solution. |
| \[Zn+CuS{{O}_{4}}\to ZnS{{O}_{4}}+Cu\] |
| \[Fe+CuS{{O}_{4}}\to FeS{{O}_{4}}+Cu\] |
| Thus [a] and [c] solution becomes colour less. |
You need to login to perform this action.
You will be redirected in
3 sec
