A) \[As{{H}_{3}}\]
B) \[N{{H}_{3}}\]
C) \[{{H}_{2}}O\]
D) \[P{{H}_{3}}\]
Correct Answer: B
Solution :
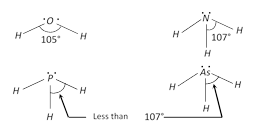 As the electronegativity of central atom decreases bond angle is decreases \[\therefore \] \[N{{H}_{3}}\]has largest bond angle.
As the electronegativity of central atom decreases bond angle is decreases \[\therefore \] \[N{{H}_{3}}\]has largest bond angle.
You need to login to perform this action.
You will be redirected in
3 sec
