Answer:
Structure of proteins The structure of proteins is much
more complex than that of any of the families of organic compounds studied.
This structural complexity arises from the fact that proteins are such large
molecules, often referred to as macromolecules. A protein may consist
of just a single strand, or it may include several strands. A protein may have
just a few a-amino acids or it may have thousands.
There are four levels of protein structure that are important in
understanding how proteins function.
The simplest two-dimensional description of
connectivity is called the primary structure. The three-dimensional
gross structure-specifically, the secondary, tertiary and quaternary
structure-describes how protein molecules are aggregated in order to carry out
their specific functions.
Primary structure The most basic aspect of protein structure
is the primary structure-a specific sequence in which the a-amino acids are
joined together by peptide bonds. Primary structure is represented using the
three-letter abbreviations for the a-amino acids.
Primary structure of insulin has two chains of insulin held
together by two disulphide (?S?S?) linkages.
[1]
Secondary structure How a protein chain is arranged
or oriented in three-dimensional space is described by secondary structure.
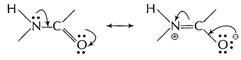
Resonance delocalisation within the amide (peptide) group
causes restricted rotation about the nitrogen-carbon bond because it
acquires partial double bond character.
S. No.
Protein
Structure-type
Number of amino acids
1.
Oxytocin
Single-stranded protein
9
![]() amino
acids
amino
acids
2.
Insulin
Two-chain protein Single-chain protein
51
![]() amino
acids 153
amino
acids 153
3.
Myoglobin
Single-chain protein
153
![]() amino
acids
amino
acids
4.
Gamma globulin
Four-chain protein
1320
![]() amino
acids
amino
acids
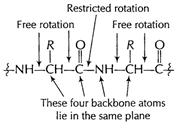
 The NH ? C = O grouping lies in a plane with bond angles
of about 120°. The carbon atoms to which the nitrogen and the carbonyl carbon
are attached (a-carbon of both amino acids) must also lie in the same plane,
Therefore, the four atoms of the peptide backbone (?C?N?C?C?) lie in a plane,
but
there are two variants.
The NH ? C = O grouping lies in a plane with bond angles
of about 120°. The carbon atoms to which the nitrogen and the carbonyl carbon
are attached (a-carbon of both amino acids) must also lie in the same plane,
Therefore, the four atoms of the peptide backbone (?C?N?C?C?) lie in a plane,
but
there are two variants.
 There is free rotation about the single bonds in a peptide.
Therefore, following are the rotational options for the peptide. The secondary
structure of a protein is generally held in place by hydrogen bonding between
the ?NH group of one amide and the C = O group of another amide. Because a
protein chain contains a large number peptide bonds, there is extensive
hydrogen bonding between peptide linkages of two separate chains or between
peptide linkages within a single chain.
There is free rotation about the single bonds in a peptide.
Therefore, following are the rotational options for the peptide. The secondary
structure of a protein is generally held in place by hydrogen bonding between
the ?NH group of one amide and the C = O group of another amide. Because a
protein chain contains a large number peptide bonds, there is extensive
hydrogen bonding between peptide linkages of two separate chains or between
peptide linkages within a single chain.
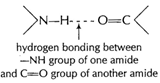 hydrogen bonding between ?NH group of one amide and C = O
group of another amide
Secondary structure can be in the form of [1]
1.
hydrogen bonding between ?NH group of one amide and C = O
group of another amide
Secondary structure can be in the form of [1]
1.![]() Helix
In which a protein chain is coiled about a central axis. It is held in place by
hydrogen bonding that occurs between the hydrogens of ?NH groups and oxygens of
C = O groups that are oriented toward each other on the inside of the helix. It
takes 3.6 amino acids to complete one turn of the helix to enable such hydrogen
bonding and a 13-member ring is formed by H-bonding. a-helix is also known as 3.6,3
helix.
2. Pleated sheet In which two protein chains are lined
up parallel to each other but running in opposite directions ?that is, the
N-terminus of one chain is near the C- terminus of the other. This puts the ?NH
group of one chain opposite the C = O group of the other chain, permitting
hydrogen bonding that holds the sheet together. Examples are given below
Helix
In which a protein chain is coiled about a central axis. It is held in place by
hydrogen bonding that occurs between the hydrogens of ?NH groups and oxygens of
C = O groups that are oriented toward each other on the inside of the helix. It
takes 3.6 amino acids to complete one turn of the helix to enable such hydrogen
bonding and a 13-member ring is formed by H-bonding. a-helix is also known as 3.6,3
helix.
2. Pleated sheet In which two protein chains are lined
up parallel to each other but running in opposite directions ?that is, the
N-terminus of one chain is near the C- terminus of the other. This puts the ?NH
group of one chain opposite the C = O group of the other chain, permitting
hydrogen bonding that holds the sheet together. Examples are given below
![]() Helix
Helix
Pleated sheet
![]() keratin
keratin
Fibroin
Myosin
Protein of silk
Actin
Dragline of silk (spider wave)
You need to login to perform this action.
You will be redirected in
3 sec
