Answer:
Acidic nature of phenol
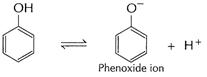 (a) Phenol reacts with Na liberating
(a) Phenol reacts with Na liberating ![]() .
.
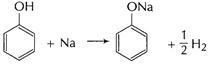 (b) Phenol reacts with NaOH (base).
(b) Phenol reacts with NaOH (base).
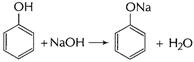 Phenol is more acidic than ethanol.
Phenol is more acidic than ethanol.
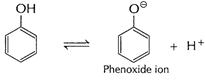 Phenoxide ion stabilised due to resonance, hence equilibrium
(i) is in towards the ionisation of the phenol.
Phenoxide ion stabilised due to resonance, hence equilibrium
(i) is in towards the ionisation of the phenol.
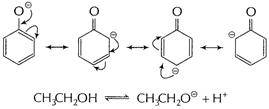
![]() shows
(l+) effect and stability of
shows
(l+) effect and stability of![]() is
decreased, makes equilibrium (ii) non-spontaneous in forward side. Thus,
is
decreased, makes equilibrium (ii) non-spontaneous in forward side. Thus, ![]() is less
acidic than phenol. [1]
is less
acidic than phenol. [1]
You need to login to perform this action.
You will be redirected in
3 sec
