Answer:
(i)
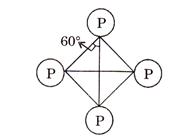
Phosphorus ![]() is more
stable than nitrogen
is more
stable than nitrogen ![]() because
of angle strain in
because
of angle strain in ![]() molecule
where the angles are only 60° rather than 109°28' (due to
tetrahedral shape).
molecule
where the angles are only 60° rather than 109°28' (due to
tetrahedral shape). ![]() Nitrogen is inert at room temperature because of high bond
enthalpy of N = N bond.
Nitrogen is inert at room temperature because of high bond
enthalpy of N = N bond. ![]()
 (ii) In vapour state sulphur exists as 83 molecule which
has two unpaired electrons in the antibonding orbitals like Og and hence
exhibit paramagnetism. 1
(iii) Nitric acid decomposes
(ii) In vapour state sulphur exists as 83 molecule which
has two unpaired electrons in the antibonding orbitals like Og and hence
exhibit paramagnetism. 1
(iii) Nitric acid decomposes ![]() which is
brown colour in which makes
which is
brown colour in which makes ![]() turns
yellow. 1
turns
yellow. 1
You need to login to perform this action.
You will be redirected in
3 sec
