Answer:
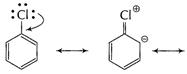
Aryl halides
are less reactive due to stability of
(C?X)
bond due to resonance effect.
(ii) more electronegativity
of C on which (X) is
attached.
(iii)
instability of phenyl carbocation
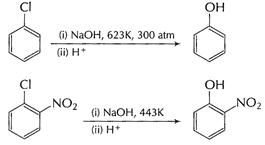
![]() reaction
is not possible. But reactivity of (C?X) bond can be increased by
(i) using
high temperature (623 K) and high pressure (300 atm).
[1]
(ii) using
electron-withdrawing group as ?
reaction
is not possible. But reactivity of (C?X) bond can be increased by
(i) using
high temperature (623 K) and high pressure (300 atm).
[1]
(ii) using
electron-withdrawing group as ?![]() ?CN,
?COOH in ortho and para-position.
?CN,
?COOH in ortho and para-position.
 (i)
(i) 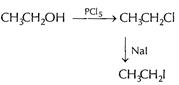 Or
[1]
(ii) (a)
Or
[1]
(ii) (a) ![]() water is
decolourised by (C=C) bond.
water is
decolourised by (C=C) bond.
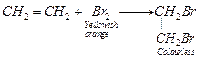 (b) Alkenes
decolourise alkaline
(b) Alkenes
decolourise alkaline ![]() (Baeyer's
reagent)
(Baeyer's
reagent)
![]()
![]() [1]
[1]
You need to login to perform this action.
You will be redirected in
3 sec
