Answer:
Melting
point of para-dichloro benzene is higher than of ortho and mete-isomers. It is
due to symmetry of para-isomers that fits in crystal lattice better as compared
to ortho and meta-isomers.
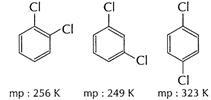 p-dichlorobenzene
has zero dipole moment and thus is non-polar. Hence, it is less soluble in
water as compared to ortho and mera-isomers. [1]
Dipole
moment of o > m >p
Also due to
symmetry of para-isomers, force of attraction is very high among molecules hence
to make soluble high energy is required. Thus solubility of para-isomers is
very low. [1]
p-dichlorobenzene
has zero dipole moment and thus is non-polar. Hence, it is less soluble in
water as compared to ortho and mera-isomers. [1]
Dipole
moment of o > m >p
Also due to
symmetry of para-isomers, force of attraction is very high among molecules hence
to make soluble high energy is required. Thus solubility of para-isomers is
very low. [1]
You need to login to perform this action.
You will be redirected in
3 sec
