Answer:
Step I
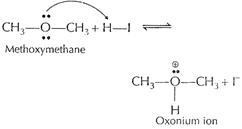 In this step ether is protonated. [1]
Step II Iodide (
In this step ether is protonated. [1]
Step II Iodide (![]() ) is a
good nucleophile. It attacks alkyl group of oxonium ion formed in step I and displaces
alcohol by
) is a
good nucleophile. It attacks alkyl group of oxonium ion formed in step I and displaces
alcohol by ![]() mechanism.
mechanism.
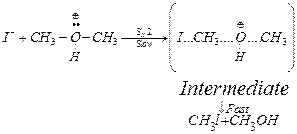 If
HI is in excess, alcohol formed further reacts with HI forming
If
HI is in excess, alcohol formed further reacts with HI forming ![]() [1]
[1]
![]()
You need to login to perform this action.
You will be redirected in
3 sec
