Answer:
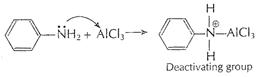
(i) Aniline does not undergo Friedel-Crafts reaction (alkylation
and acetylation) as it forms salt with AICI3 (catalyst, Lewis acid) ?![]() group (electron-pair
donor, Lewis base) acquires positive charge and thus -
group (electron-pair
donor, Lewis base) acquires positive charge and thus -![]() group in
aniline becomes deactivating group for further reaction.
group in
aniline becomes deactivating group for further reaction.
 [1]
(ii)
[1]
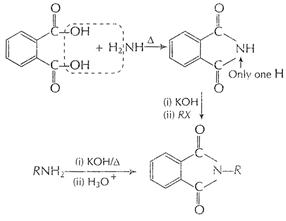
(ii) 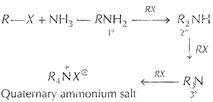 In the above method, when R?X reacts with NH3 1°,
2°, 3° amines and quaternary ammonium salt is formed. In Gabriel phthalimide
reaction, two H of NH3 are blocked by phthalimide formation (only one H is left
reactive), hence 1° amine is formed.
In the above method, when R?X reacts with NH3 1°,
2°, 3° amines and quaternary ammonium salt is formed. In Gabriel phthalimide
reaction, two H of NH3 are blocked by phthalimide formation (only one H is left
reactive), hence 1° amine is formed.
 [1]
[1]
You need to login to perform this action.
You will be redirected in
3 sec
