A) 4
B) 6
C) 5
D) 9
Correct Answer: B
Solution :
| For bubble A: |
| If \[{{P}_{A}}\] is the pressure inside the bubble then |
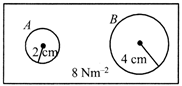 |
| \[{{P}_{A}}-8=\frac{4T}{{{R}_{A}}}=\frac{4\times 0.04}{0.02}=8\,\,\Rightarrow {{P}_{A}}=16N/{{m}^{2}}\] |
| According to ideal gas equation, |
| \[{{P}_{A}}{{V}_{A}}={{n}_{A}}R{{T}_{A}}\,\,\Rightarrow \,\,16\times \frac{4}{3}\pi {{(0.02)}^{3}}={{n}_{A}}R{{T}_{A}}\] ...(i) |
| For bubble B: |
| If \[{{P}_{B}}\] is the pressure inside the bubble then |
| \[{{P}_{B}}-8=\frac{4T}{{{R}_{B}}}=\frac{4\times 0.04}{0.04}=4\] \[\Rightarrow \,\,{{P}_{B}}=12N/{{m}^{2}}\] |
| According to ideal gas equation, |
| \[{{P}_{B}}{{V}_{B}}={{n}_{B}}R{{T}_{B}}\Rightarrow 12\times \frac{4}{3}\pi {{(0.04)}^{3}}={{n}_{B}}R{{T}_{B}}\] ...(ii) |
| Dividing (ii) by (i) we get |
| \[\frac{12\times \frac{4}{3}\pi {{(0.04)}^{3}}}{16\times \frac{4}{3}\pi {{(0.02)}^{3}}}=\frac{{{n}_{B}}}{{{n}_{A}}}\] \[\left[ \because \,\,{{T}_{A}}={{T}_{B}} \right]\] |
| \[\therefore \,\,\,\frac{{{n}_{B}}}{{{n}_{A}}}=6\] |
You need to login to perform this action.
You will be redirected in
3 sec
