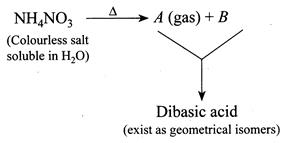
 The gas and dibasic acid are, respectively,
The gas and dibasic acid are, respectively,
A) \[N{{O}_{2}},\,{{H}_{2}}{{N}_{2}}{{O}_{2}}\]
B) \[{{N}_{2}}O,\,{{H}_{2}}{{N}_{2}}{{O}_{2}}\]
C) \[{{N}_{2}}\,{{H}_{2}}{{N}_{2}}O\]
D) \[NO,\,\,{{H}_{2}}{{N}_{2}}{{O}_{3}}\]
Correct Answer: B
Solution :
[b] \[N{{H}_{4}}N{{O}_{3}}\xrightarrow{\Delta }\underset{(A)\,(gas)}{\mathop{{{N}_{2}}O}}\,+\underset{(B)}{\mathop{2{{H}_{2}}O}}\,\] \[\underset{(A)}{\mathop{{{N}_{2}}O}}\,+\underset{(B)}{\mathop{{{H}_{2}}O}}\,\xrightarrow{{}}\underset{\text{Hypointrous acid}}{\mathop{{{H}_{2}}{{N}_{2}}{{O}_{2}}}}\,\]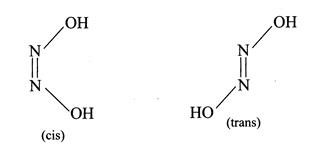
You need to login to perform this action.
You will be redirected in
3 sec
