| (I) \[PHC{{H}_{2}}C{{H}_{2}}Br\] and \[PhC{{D}_{2}}C{{H}_{2}}Br\] towards dehydrohalogenation by strong base. |
(II) Conversion of: 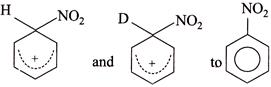 |
(III) Conversion of: 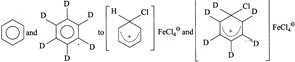 |
(IV) Conversion of benzene to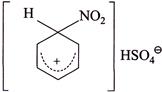 |
A) I, II
B) I, II, III
C) II, III, IV
D) III, IV
Correct Answer: A
Solution :
[a] For \[1{}^\circ \] isotope effect \[{{K}_{H}}/{{K}_{D}}>1\] For \[2{}^\circ \] isotope effect \[{{K}_{H}}/{{K}_{D}}<1\] Since both are \[1{}^\circ RX,\] so dehydrohalogenation reaction proceeds via \[E2\] mechanism and this depends on the breaking of the \[(C-H)\] or \[(C-D)\]bond. It shows \[1{}^\circ \] isotope effect.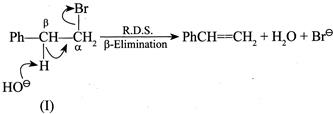
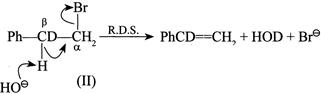 Since in reaction (I) and \[(C-H)\] bond is broken and in (II) the \[(C-D)\] bond is broken, the \[(C-H)\] bond is broken more easily than the \[(C-D)\] bond. So the rate of (I) is faster than that of (II). Hence, \[{{K}_{H}}/{{K}_{D}}>1\].
Since in reaction (I) and \[(C-H)\] bond is broken and in (II) the \[(C-D)\] bond is broken, the \[(C-H)\] bond is broken more easily than the \[(C-D)\] bond. So the rate of (I) is faster than that of (II). Hence, \[{{K}_{H}}/{{K}_{D}}>1\]. 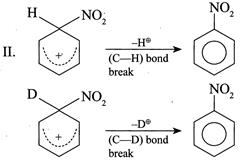 So, \[{{K}_{H}}/{{K}_{D}}>1\]
So, \[{{K}_{H}}/{{K}_{D}}>1\] 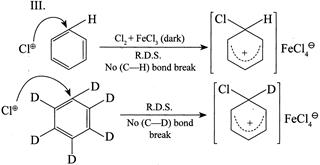 In both the reactions neither the \[(C-H)\] nor the \[(C-D)\] bond break and rate of both the reaction is: Rate \[=K\,[C{{l}^{\oplus }}]\] [Benzene]; so \[{{K}_{H}}/{{K}_{D}}=1.\] IV. In the nitration to intermediate no \[(C-H)\] bond is broken in R.D.S. \[\therefore \,\,\,\,{{K}_{H}}/{{K}_{D}}=1\]
In both the reactions neither the \[(C-H)\] nor the \[(C-D)\] bond break and rate of both the reaction is: Rate \[=K\,[C{{l}^{\oplus }}]\] [Benzene]; so \[{{K}_{H}}/{{K}_{D}}=1.\] IV. In the nitration to intermediate no \[(C-H)\] bond is broken in R.D.S. \[\therefore \,\,\,\,{{K}_{H}}/{{K}_{D}}=1\]
You need to login to perform this action.
You will be redirected in
3 sec
