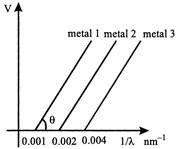
A) Ratio of work functions \[{{\phi }_{1}}:{{\phi }_{2}}:{{\phi }_{3}}=\,\,1:2:5\]
B) Ratio of work functions \[{{\phi }_{1}}:{{\phi }_{2}}:{{\phi }_{3}}=\,\,4:2:1\]
C) tan 6 is directly proportional to hc/e, where h is Planck?s constant and c is the speed of light.
D) The violet colour light can eject photoelectrons from metals 2 and 3.
Correct Answer: C
Solution :
\[\frac{hc}{e\lambda }-\phi =eV\] \[V=\frac{hc}{e\lambda }-\frac{\phi }{e}\] For plate 1: Plate 2 Plate 3 \[\frac{{{\phi }_{1}}}{hc}=0.001\,\,\,\,\,\,\,\,\,\,\frac{{{\phi }_{1}}}{hc}=0.001\,\,\,\,\,\,\,\,\,\,\,\frac{{{\phi }_{1}}}{hc}=0.001\] \[{{\phi }_{1}}:{{\phi }_{2}}:{{\phi }_{3}}\,\,=\,\,1:2:4\] For plate 2, threshold wavelength \[\lambda =\frac{hc}{{{\phi }_{2}}}-=\frac{hc}{0.002hc}=\frac{1000}{2}=500\,nm\] For plate 3, threshold wavelength \[\lambda =\frac{hc}{{{\phi }_{3}}}\,\,-=\frac{hc}{0.004\,hc}=\frac{1000}{2}=250\,nm\] since violet colour light \[\lambda \] is 400 nm, so \[{{\lambda }_{violet}}<{{\lambda }_{threshold}}\] so, violet colour light will eject photo-electrons from plate 2 and not plate 3.You need to login to perform this action.
You will be redirected in
3 sec
