| (I) |
| (II) |
| (III) |
| (IV) |
A) \[I>IV>III>II\]
B) \[II>III>IV>I\]
C) \[I>III>II>IV\]
D) \[I>IV>II>III\]
Correct Answer: A
Solution :
[a] 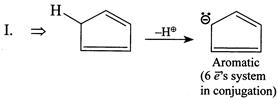 |
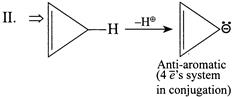 |
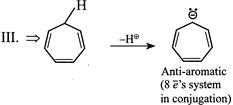 |
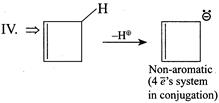 |
You need to login to perform this action.
You will be redirected in
3 sec
