A) \[C{{r}^{3+}}\]
B) \[M{{n}^{3+}}\]
C) \[F{{e}^{2+}}\]
D) \[C{{o}^{2+}}\]
Correct Answer: B
Solution :
[b] \[\mu =4.9\] BM corresponds to \[n=4\]and for \[\mu =2.80\] BM corresponds to\[n=2\]. For \[{{d}^{4}}\] configuration, in a weak fields ligands it will have 4 unpaired electrons. In strong field ligand, \[{{d}^{4}}\]configuration will have one paired electrons and two unpaired electrons. Note:- \[{{d}^{1}}\] to \[{{d}^{3}}\] configuration, no pairing will occur whether the ligand field is weak or strong. But for \[{{d}^{4}}\]configuration only one pairing will occur in strong field ligands Therefore, \[M{{n}^{3+}}\] have \[3{{d}^{4}}\] configuraton. In weak field ligands: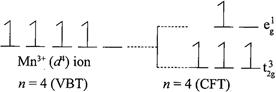 In strong field ligands:
In strong field ligands: 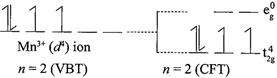
You need to login to perform this action.
You will be redirected in
3 sec
