A) \[{{\left[ Ni{{\left( CN \right)}_{4}} \right]}^{-2}}\] and \[\left[ Ni{{\left( CO \right)}_{4}} \right]\] have the same magnetic moment
B) \[{{\left[ NiC{{l}_{4}} \right]}^{-2}}\] and \[{{[PtC{{l}_{4}}]}^{-2}}\] have different shape.
C) Hybrid state of Co in \[{{\left[ Co{{\left( Ox \right)}_{3}} \right]}^{-3}}\] is \[s{{p}^{3}}{{d}^{2}}\]
D) In brown-ring complex \[\left[ Fe{{\left( {{H}_{2}}O \right)}_{5}}NO \right]S{{O}_{4}}\] oxidation state of Fe is +1
Correct Answer: C
Solution :
[c] \[{{\left[ CO{{(ox)}_{3}} \right]}^{-3}}\] \[C{{o}^{+3}}=\left[ Ar \right]4{{s}^{0}}3{{d}^{6}}\]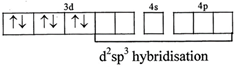
You need to login to perform this action.
You will be redirected in
3 sec
