| I. t-Butylamine |
| II. Neopentylamine |
| III. Diethylamine |
| IV. p-Toluidine |
| V. m-Nitroaniline |
| VI. Vinylamine |
| VII. p-Nitroaniline |
| VIII. Allylamine |
A) 3
B) 4
C) 5
D) 6
Correct Answer: D
Solution :
[d] Halides or tosylates which are unreactive in \[S{{N}^{2}}\]cannot be used.| (I) to (VI) cannot be prepared but (VII) and (VIII) can be prepared. |
| (I) It cannot be prepared since \[M{{e}_{3}}C-X\]is \[3{}^\circ \]halide. |
| (II) It cannot be prepared since \[M{{e}_{3}}C-C{{H}_{2}}-X\]is neopentyl halide and does not undergo \[S{{N}^{2}}\]reaction due to steric hindrance. |
| (III) It cannot be prepared since \[2{}^\circ \] amine gives poor yield due to elimination. |
(IV) It cannot be prepared, since arylhalide, 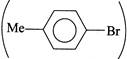 is not activated by EDG (Me group). \[ArSN\]reaction takes place only when the benzene ring is activated by EWG present at o- and p-positions. is not activated by EDG (Me group). \[ArSN\]reaction takes place only when the benzene ring is activated by EWG present at o- and p-positions. |
(V) It cannot be prepared, since EWG (\[-N{{O}_{2}}\] group) 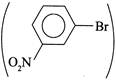 is present at w-position. \[ArSN\]reaction does not take place. is present at w-position. \[ArSN\]reaction does not take place. |
| (VI) It cannot be prepared, since vinyl halides\[(C{{H}_{2}}=\,=CH-X)\] are unreactive in \[S{{N}^{2}}\]reactions |
(VII) It can be prepared, since benzene ring 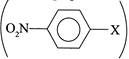 is activated by EWG (\[-N{{O}_{2}}\] group) at p-position and hence \[ArSN\] reaction takes place. is activated by EWG (\[-N{{O}_{2}}\] group) at p-position and hence \[ArSN\] reaction takes place. |
| (VIII) It can be prepared, since allyl halides\[(C{{H}_{2}}=\,=CHC{{H}_{2}}X)\] can undergo \[S{{N}^{2}}\] reaction. |
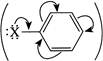 . But, containing EWG (\[\bar{e}\]withdrawing groups) (e.g., \[-N{{O}_{2}}\]) at o-and p-position activates the benzene ring for \[ArSN\]reaction. That is why o- and p-nitroaniline can be prepared but \[ArX\]containing EDG (\[\bar{e}\]-donating groups) cannot be used for the preparation of corresponding amines.
. But, containing EWG (\[\bar{e}\]withdrawing groups) (e.g., \[-N{{O}_{2}}\]) at o-and p-position activates the benzene ring for \[ArSN\]reaction. That is why o- and p-nitroaniline can be prepared but \[ArX\]containing EDG (\[\bar{e}\]-donating groups) cannot be used for the preparation of corresponding amines.
You need to login to perform this action.
You will be redirected in
3 sec
