A) Compressing the gas isothermally will require morc work to be done.
B) Compressing the gas through adiabatic process will require more work to be done.
C) Compr essing the gas isothermally or adiabatically will require the same amount of work.
D) Which of the case (whether compression through isothermal or through adiabatic process) requires more work will depend upon the atomicity of the gas.
Correct Answer: B
Solution :
\[{{W}_{ext}}=\] negative of area with volume-axis \[W\text{ }\left( adiabatic \right)>W\left( isothermal \right)\]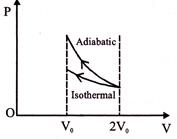
You need to login to perform this action.
You will be redirected in
3 sec
