A) \[N{{a}_{2}}[{{B}_{4}}{{O}_{5}}{{(OH)}_{4}}].8{{H}_{2}}O\]
B) \[2NaB{{O}_{2}}.N{{a}_{2}}{{B}_{2}}{{O}_{3}}\cdot 10{{H}_{2}}O\]
C) \[N{{a}_{2}}[{{B}_{4}}{{({{H}_{2}}O)}_{4}}{{O}_{7}}]\cdot 6{{H}_{2}}O\]
D) None of these
Correct Answer: A
Solution :
Idea This problem is based on molecular formula representation and structure of borax, students are advised to go through the structure of borax and their molecular formula representation to solve the question. Borax molecule is actually made of two tetrahedral and two triangular units joined as follows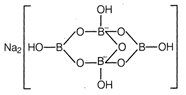 So, correct representation of borax is \[N{{a}_{2}}[{{B}_{4}}{{O}_{5}}{{(OH)}_{4}}]\cdot 8{{H}_{2}}O\] TEST Edge These types of questions are asked in exam to judge the knowledge of student on structure of a various inorganic molecules, students are advised to go through the understanding of structure of various inorganic compounds. Question related to structure of compounds like\[{{P}_{4}}{{O}_{10}}\],\[{{P}_{4}}{{O}_{6}}\]etc., may also be asked.
So, correct representation of borax is \[N{{a}_{2}}[{{B}_{4}}{{O}_{5}}{{(OH)}_{4}}]\cdot 8{{H}_{2}}O\] TEST Edge These types of questions are asked in exam to judge the knowledge of student on structure of a various inorganic molecules, students are advised to go through the understanding of structure of various inorganic compounds. Question related to structure of compounds like\[{{P}_{4}}{{O}_{10}}\],\[{{P}_{4}}{{O}_{6}}\]etc., may also be asked.
You need to login to perform this action.
You will be redirected in
3 sec
