A) \[\text{5}\,\text{kJ}\]
B) \[\text{25}\,\text{kJ}\]
C) \[\text{15}\,\text{kJ}\]
D) \[20\,\text{kJ}\]
Correct Answer: C
Solution :
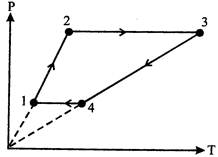
\[\text{Proces}{{\text{s}}_{1\to 2}}\]and\[\text{Proces}{{\text{s}}_{3\to 4}}\]are isochoric processes. \[{{W}_{12}}=0,\,\,{{W}_{34}}=0,\,\,{{W}_{23}}=nR({{T}_{3}}-{{T}_{2}})=3R(1600-400)\] \[=3600R\] \[{{W}_{41}}=nR({{T}_{1}}-{{T}_{4}})=3R(200-800)=-1800R\] \[W=(3600-1800)R=1800R=15\,\text{kJ}\].You need to login to perform this action.
You will be redirected in
3 sec
