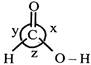
A) \[x>y>z\]
B) \[x<y<z\]
C) \[x=y>z\]
D) Can't be predicted
Correct Answer: A
Solution :
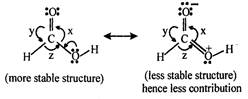 Therefore, Repulsion between both C-O bonds will be greater than the repulsion between C - H bond and C - OH bond or between C - H and C = O bond. Repulsion between C - H and C = O bond is greater than that of between C - H and C - OH bond, because double bond character in C - OH bond will always be less than C = O.
Therefore, Repulsion between both C-O bonds will be greater than the repulsion between C - H bond and C - OH bond or between C - H and C = O bond. Repulsion between C - H and C = O bond is greater than that of between C - H and C - OH bond, because double bond character in C - OH bond will always be less than C = O.
You need to login to perform this action.
You will be redirected in
3 sec
