A)
![]()
B)
![]()
C)
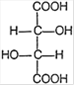
D)
![]()
Correct Answer: D
Solution :
Structure [a] shows geometrical isomerism because of the presence of different groups at double bonded carbon atoms. Structure [b] and [c] show optical isomerism because of the absence of symmetry elements. Structure [d] shows neither geometrical nor optical isomerism as it does not satisfy the conditions for optical and geometrical isomerism.You need to login to perform this action.
You will be redirected in
3 sec
