| Statement I: Detection of chlorine in 2, 4, 6 -trinitrochlorobenzene can be done directly by addition of aq. \[AgN{{O}_{3}}\]solution. |
| Statement II: \[C-Cl\] bond is weakened by electron withdrawing - \[N{{O}_{2}}\] group. |
A) Statement I is true; Statement II is true; Statement II is not a correct explanation for Statement I.
B) Statement I is true; Statement II is false.
C) Statement I is false; Statement II is true.
D) Statement I is true; Statement II is true; Statement II is not a correct explanation for Statement I.
Correct Answer: D
Solution :
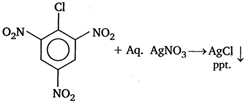 2, 4, 6- trinitrochlorobenzene The presence of electron withdrawing group (like\[-N{{O}_{2}}\]) makes the nucleophilic aromatic substitution easier, as it decreases the strength of \[C-Cl\] bond. Thus, \[-Cl\] gives ppt. of \[AgCl\] with aq.\[AgN{{O}_{3}}\].
2, 4, 6- trinitrochlorobenzene The presence of electron withdrawing group (like\[-N{{O}_{2}}\]) makes the nucleophilic aromatic substitution easier, as it decreases the strength of \[C-Cl\] bond. Thus, \[-Cl\] gives ppt. of \[AgCl\] with aq.\[AgN{{O}_{3}}\].
You need to login to perform this action.
You will be redirected in
3 sec
