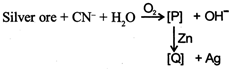 Identify the complexes [P] and [Q]
Identify the complexes [P] and [Q]
A) \[P={{[Ag{{(CN)}_{4}}]}^{-}},Q,={{[Zn{{(CN)}_{4}}]}^{2-}}\]
B) \[P={{[Ag{{(CN)}_{4}}]}^{-}},Q,={{[Zn{{(CN)}_{6}}]}^{4-}}\]
C) \[P={{[Ag{{(CN)}_{4}}]}^{3-}},Q,={{[Zn{{(CN)}_{4}}]}^{2-}}\]
D) \[P={{[Ag{{(CN)}_{2}}]}^{-}},Q,={{[Zn{{(CN)}_{4}}]}^{2-}}\]
Correct Answer: D
Solution :
\[2Ag+4C{{N}^{-}}\,+{{H}_{2}}O\,+\frac{1}{2}{{O}_{2}}\xrightarrow{\,}2{{[Ag{{(CN)}_{2}}]}^{-}}+2O{{H}^{-}}\] \[2{{[Ag{{(CN)}_{2}}]}^{-}}+Zn\,\xrightarrow{\,}\,{{[Zn\,{{(CN)}_{4}}]}^{2-}}+2Ag\].You need to login to perform this action.
You will be redirected in
3 sec
