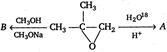
 A and B are
A and B are
A) \[\text{C}{{\text{H}}_{\text{3}}}-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ _{\text{18}}\text{OH} \end{smallmatrix}}{\overset{\begin{smallmatrix} \text{C}{{\text{H}}_{\text{3}}} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{OH} \end{smallmatrix}}{\mathop{\text{C}{{\text{H}}_{\text{2}}}\text{,}}}\,\text{C}{{\text{H}}_{\text{3}}}-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{OH} \end{smallmatrix}}{\overset{\begin{smallmatrix} \text{C}{{\text{H}}_{\text{3}}} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{OC}{{\text{H}}_{\text{3}}} \end{smallmatrix}}{\mathop{\text{C}{{\text{H}}_{\text{2}}}}}\,\]
B) \[\text{C}{{\text{H}}_{\text{3}}}-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{OH} \end{smallmatrix}}{\overset{\begin{smallmatrix} \text{C}{{\text{H}}_{\text{3}}} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}}\,---\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ _{\text{18}}\text{OH} \end{smallmatrix}}{\mathop{\text{C}{{\text{H}}_{\text{2}}}\text{,}}}\,\text{C}{{\text{H}}_{\text{3}}}-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{OH} \end{smallmatrix}}{\overset{\begin{smallmatrix} \text{C}{{\text{H}}_{\text{3}}} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{OC}{{\text{H}}_{\text{3}}} \end{smallmatrix}}{\mathop{\text{C}{{\text{H}}_{\text{2}}}}}\,\]
C) Both [a] and [b] are correct
D) None of the above is correct
Correct Answer: A
Solution :
A in the acidic medium, appears as \[{{S}_{N}}1\] type product as \[3{}^\circ \] carbocation is more stable that's why \[C{{H}_{3}}-\underset{+}{\mathop{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}\,}}\,-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C{{H}_{2}}}}\,\]forms and A is \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ 18OH \end{smallmatrix}}{\mathop{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}\,}}\,-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C{{H}_{2}}}}\,\] B appears as \[{{S}_{N}}2\] type product as \[1{}^\circ \] is les sterically hindered that's why \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}\,}}\,-\underset{+}{\mathop{C{{H}_{2}}}}\,\]forms and B is \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}\,}}\,-\underset{\begin{smallmatrix} | \\ OC{{H}_{3}} \end{smallmatrix}}{\mathop{C{{H}_{2}}}}\,\]You need to login to perform this action.
You will be redirected in
3 sec
