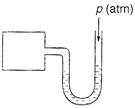
A) 9 cm Hg
B) 18 cm Hg
C) 27 cm Hg
D) None of these
Correct Answer: C
Solution :
\[\therefore \] \[{{x}_{2}}=\frac{{{m}_{1}}}{{{m}_{2}}}{{x}_{1}}\] \[{{m}_{1}}=\frac{M}{4},\,{{x}_{1}}=\frac{R}{2}\] \[{{m}_{2}}=\frac{3M}{4}\] \[{{x}_{2}}=-\frac{M/4}{3M/4}\cdot \frac{R}{2}=\frac{-R}{6}\] \[\frac{R}{6}\] \[{{d}_{w}}\] \[{{d}_{o}}\] of HgYou need to login to perform this action.
You will be redirected in
3 sec
