A) \[{{[Fe{{(CN)}_{6}}]}^{4-}}\]
B) \[{{[Fe{{({{H}_{2}}O)}_{6}}]}^{2+}}\]
C) \[{{[Cu{{(N{{H}_{3}})}_{6}}]}^{2+}}\]
D) \[{{[Ni{{(CN)}_{6}}]}^{4-}}\]
Correct Answer: A
Solution :
In \[{{[Fe{{(CN)}_{6}}]}^{4-}},\,Fe\], is in +2 oxidation state. \[_{26}Fe-1{{s}^{2}},\,2{{s}^{2}}\,2{{p}^{6}},\,3{{s}^{2}}s{{p}^{6}},\,4{{s}^{2}}\]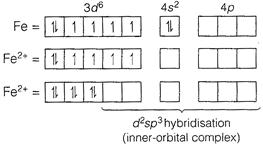 (\[C{{N}^{-}}\] is a strong field ligand and causes pairing of electrons.) Therefore, \[{{[Fe{{(CN)}_{6}}]}^{4-}}\] complex ion is inner-orbital and diamagnetic (as all electrons are paired).
(\[C{{N}^{-}}\] is a strong field ligand and causes pairing of electrons.) Therefore, \[{{[Fe{{(CN)}_{6}}]}^{4-}}\] complex ion is inner-orbital and diamagnetic (as all electrons are paired).
You need to login to perform this action.
You will be redirected in
3 sec
