| Statement 1: The temperature of gas continuously increases. |
| Statement 2: According to first law of thermodynamics \[dQ=dU+dW\]where symbols have their usual meaning. |
A) Statement -1 false, Statement-2 is true.
B) Statement-1 is true, Statement-2 is true; Statement-2 is a correct explanation for Statement-1
C) Statement-1 is true, Statement-2 is true; Statement-2 is not a correct explanation for Statement-1
D) Statement -1 true, Statement-2 is false.
Correct Answer: A
Solution :
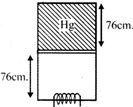
Initial condition Volume = 76cm x A Pressure = (76 + 76) dg = 152dg Final condition Volume= 152cm x A Pressure = 76dg \[\therefore \] \[{{P}_{1}}{{V}_{1}}={{P}_{2}}{{V}_{2}}\] \[\therefore \]Both points lie on the same isothermal line i.e. both have same T.You need to login to perform this action.
You will be redirected in
3 sec
