A) All \[I-Cl\] bonds are equivalent
B) Molecule is polar and non-planar
C) All adjacent bond angles are equal
D) All hybrid orbital?s of central atom having equal s-character
Correct Answer: C
Solution :
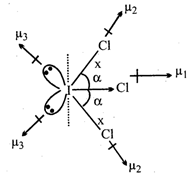 \[({{\mu }_{D}}\ne 0)\]\[x>y\] Molecule is polar and planar. Both \[\angle ClICl\]are equal Equatorial \[I-Cl\]bond has more s-character than axial \[I-Cl\]bond.
\[({{\mu }_{D}}\ne 0)\]\[x>y\] Molecule is polar and planar. Both \[\angle ClICl\]are equal Equatorial \[I-Cl\]bond has more s-character than axial \[I-Cl\]bond.
You need to login to perform this action.
You will be redirected in
3 sec
