| A piston cylinder contains a liquid vapour mixture of water at 300 K. During a constant vapour process, 750 kJ of heat is transferred to water. |
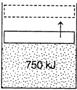 |
| Now, choose the correct option. |
A) Entropy change of water in process is \[2.5\,kJ/K\]
B) Entropy change of water in process is \[-\,2.5\,kJ/K\]
C) Entropy change of water is zero
D) Entropy change of system is \[2.5\,kJ/K\]
Correct Answer: A
Solution :
System undergoes an internally reversible isothermal process. So, entropy change of system is \[\Delta S=\frac{Q}{{{T}_{system}}}=\frac{750}{300}=2.5\frac{kJ}{K}\]You need to login to perform this action.
You will be redirected in
3 sec
