A) 3
B) 4
C) 1
D) 2
Correct Answer: D
Solution :
| 2-methylbutane has 4 types of hydrogen atoms. |
| \[{{\operatorname{CH}}_{3}}-\underset{{}}{\overset{\overset{{}}{\mathop \underset{|}{\mathop C{{H}_{3}}}\,}\,}{\mathop{CH}}}\,-C{{H}_{2}}C{{H}_{3}}\] |
| Hence, on monochlorination, it gives 4 product. |
| (i) |
| |
| It will exist in from of enantiomeric pair. |
| (ii) |
 |
| It has no chiral carbon, so no enaniomerism possible. |
| (iii) |
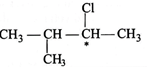 |
| It has no chiral carbon atom, so no enantiomerism. |
You need to login to perform this action.
You will be redirected in
3 sec
