| A gas undergoes two processes as shown below. |
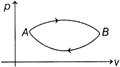 |
| Ratio of molar heat capacities for process AB and BA is |
A) less than 1
B) greater than 1
C) equal to 1
D) Data insufficient
Correct Answer: B
Solution :
We have, \[C=\frac{\Delta Q}{\Delta T}=\frac{\Delta U+\Delta W}{\Delta T}\]For paths A and B, \[\Delta {{U}_{1}}=\Delta {{U}_{2}}\]and \[\Delta {{W}_{2}}=\Delta {{W}_{1}}\] So, \[{{C}_{2}}>{{C}_{1}}\]\[\Rightarrow \]\[\frac{{{C}_{2}}}{{{C}_{1}}}>1\]You need to login to perform this action.
You will be redirected in
3 sec
