| The correct order of increasing basicity of given compound is |
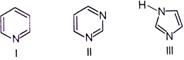 |
A) \[II<III<I\]
B) \[I<II<III\]
C) \[II<I<III\]
D) \[III<II<I\]
Correct Answer: C
Solution :
Among the given compounds, III is most basic as one of the nitrogen atom is \[s{{p}^{3}}\text{-}\]hybridised. Between I and II, I is more basic as the lone pair on N atom is relatively more available for donation as compared to II. Also, in compound II, the other N-atom shows -I-effect which makes it a weaker base. Thus, the correct increasing order of basicity is II < I < III.You need to login to perform this action.
You will be redirected in
3 sec
