A) \[s{{p}^{3}}\]and \[{{d}^{3}}s{{p}^{2}}\]
B) \[d\,s{{p}^{2}}\] and \[{{d}^{2}}s{{p}^{3}}\]
C) \[s{{p}^{3}}\]and \[{{d}^{2}}s{{p}^{3}}\]
D) \[d\,s{{p}^{2}}\]and \[s{{p}^{3}}{{d}^{2}}\]
Correct Answer: C
Solution :
| \[Ni\,{{(CN)}_{4}}Ni:[Ar]\,3{{d}^{8}}4{{s}^{2}}=t_{2g}^{2,\,2,\,2}e_{g}^{2,\,2}\] |
| \[C{{N}^{-}}\]is a strong filed ligand, hence it will cause pairing of electrons. Hence, \[ds{{p}^{2}}.\] |
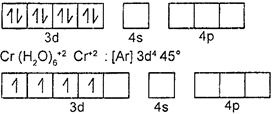 |
| octahedral complex: \[{{d}^{2}}s{{p}^{3}}\] |
You need to login to perform this action.
You will be redirected in
3 sec
