A) \[C{{O}_{2}}\]
B) \[C{{H}_{4}}\]
C) \[N{{H}_{3}}\]
D) \[N{{F}_{3}}\]
Correct Answer: C
Solution :
| \[C{{O}_{2}}\] and \[C{{H}_{4}}\] have zero dipole moment as these are symmetrical in nature. Between \[N{{H}_{3}}\] and \[N{{F}_{3}}\], \[N{{H}_{3}}\] has greater dipole moment, though in \[N{{H}_{3}}\] and \[N{{F}_{3}}\] both, N possesses one lone pair of electrons. This is because in case of \[N{{H}_{3}}\] the net \[N-H\] bond dipole is in the same direction as the direction of dipole of lone pair, but in case ofNF3, the direction of net bond dipole moment of three \[-N-F\] bonds is opposite to that of the dipole moment of the lone pair. | |
[a] 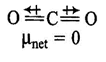 | [b] 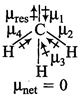 |
[c] 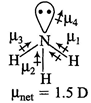 | [d] 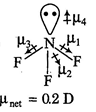 |
You need to login to perform this action.
You will be redirected in
3 sec
