A) 1, 1-dichloropropane
B) 1, 2-dichloropropane
C) 2, 2-dichloropropane
D) 1, 3-dichloropropane
Correct Answer: C
Solution :
\[{{C}_{3}}{{H}_{6}}O\](carbonyl compound) gives iodoform test (yellow ppt. with \[{{I}_{2}}/NaOH\]) This suggest that, it contains \[-\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\]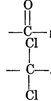 group is due to hydrolysis of Then \[{{C}_{3}}{{H}_{6}}C{{l}_{2}}\] will be group is due to hydrolysis of Then \[{{C}_{3}}{{H}_{6}}C{{l}_{2}}\] will be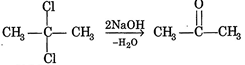 2, 2 dichloropropane 2, 2 dichloropropane |
You need to login to perform this action.
You will be redirected in
3 sec
