A) \[{{H}_{3}}{{O}^{+}},Mn{{O}_{4}}^{-}/{{H}^{+}}\]
B) \[{{H}_{3}}{{O}^{+}},Cr{{O}_{3}}/pyridine\]
C) \[B{{H}_{3}}.THF/{{H}_{2}}{{O}_{2}},Cr{{O}_{3}}/pyridine\]
D) \[B{{H}_{3}}.THF/{{H}_{2}}{{O}_{2}},O{{H}^{-}},Cr{{O}^{3}}/{{H}_{3}}{{O}^{+}}\]
Correct Answer: C
Solution :
| |
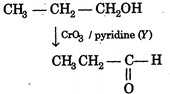 |
| "Propene undergoes hydroboration in presence of peroxide, to give propanol. This formed alcohol undergoes oxidation with oxidizing agent \[Cr{{O}_{3}}\] in pyridine to give propanol. |
You need to login to perform this action.
You will be redirected in
3 sec
