A) \[{{\left( N{{H}_{4}} \right)}_{2}}C{{r}_{2}}{{O}_{7}}\xrightarrow{\Delta }\]
B) \[N{{H}_{4}}CL+NaN{{O}_{2}}\xrightarrow{\Delta }\]
C) \[N{{H}_{3}}+NaN{{O}_{2}}\xrightarrow{\Delta }\]
D) \[{{N}_{2}}O+Cu\xrightarrow{\Delta }\]
Correct Answer: B
Solution :
| In laboratory, pure \[{{N}_{2}}\] gas is prepared by heating \[N{{H}_{4}}Cl\] with sodium nitrite. |
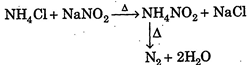 |
| \[{{\left( N{{H}_{4}} \right)}_{2}}C{{r}_{2}}{{O}_{7}}\xrightarrow{\Delta }{{N}_{2}}+Cr2{{O}_{3}}+4{{H}_{2}}O\] |
| This reaction will not give pure \[{{N}_{3}}\] as it contain traces of \[C{{r}_{2}}{{O}_{3}}\]. |
You need to login to perform this action.
You will be redirected in
3 sec
