| The following mechanism has been proposed for the exothermic catalyzed complex reaction |
| \[\operatorname{A}+BIAB\xrightarrow{{{k}_{1}}}\operatorname{AB}+I\xrightarrow{{{k}_{2}}}\operatorname{P}+A\] |
| If \[{{k}_{1}}\], is much smaller than \[{{\operatorname{k}}_{2}}\] the most suitable qualitative plot of potential energy (P. E.) versus reaction co-ordinate (R.C.) for the above reaction |
A)
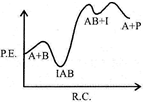
B)
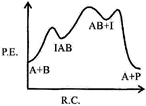
C)
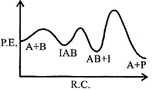
D)
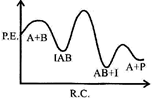
Correct Answer: B
Solution :
| \[A+B\operatorname{IAB};\] So \[{{E}_{a}}(f)\]is high and fast |
| \[{{\operatorname{E}}_{a}}\][b] is low.\[{{k}_{1}}<<{{k}_{2}};\] So, \[{{\operatorname{E}}_{a}}\]for this step is very high and for next step is low and overall reaction is exothermic. |
You need to login to perform this action.
You will be redirected in
3 sec
