| Based on the given diagram, the correct statements regarding the homogenous solutions of two volatile liquids is |
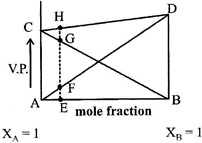 |
| (1) Plots AD and BC show that Raoult's law is obeyed for the solution in which B is a solvent and A is the solute and as well as for that in which A is solvent and B is solute. |
| (2) Plot CD shows that Dalton's law of partial pressures is obeyed by the binary solution of components A and B. |
| (3) EF + EG = EH; and AC and BD correspond to the vapour pressures of the pure solvents A and B respectively. |
A) Only 1
B) 2 and 3
C) 1 and 3
D) All Statements are correct
Correct Answer: D
Solution :
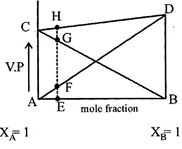 |
| Plots AD show vapour pressure of B containing A volatile component. Plot BC show vapour pressure of A containing B volatile component. Plot CD show vapour pressure of liquid solution containing A and B volatile components. |
| \[P={{P}_{A}}+{{P}_{B}}\] |
| \[\therefore \] \[{{E}_{H}}=EF+EG\] |
You need to login to perform this action.
You will be redirected in
3 sec
