A) \[CHC{{l}_{3}}\]
B)
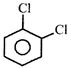
C)
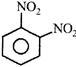
D)
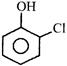
Correct Answer: D
Solution :
[d]| Due to steric repulsion among \[Cl\]-atoms observed \[\angle Cl\text{ }C\text{ }Cl\] is found to be greater |
| Than expected. |
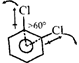 |
| Bond angle between both C-\[Cl\] bonds is greater than \[60{}^\circ \] due to steric repulsion between both \[Cl\] - atoms, hence \[{{\mu }_{D}}\](observed) \[<{{\mu }_{D}}\](Theoretical) |
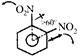 Observed bond angle between both\[CN{{O}_{2}}\]bonds is found to be greater than \[60{}^\circ \]due to Steric repulsion, between both\[N{{O}_{2}}\], groups, Observed bond angle between both\[CN{{O}_{2}}\]bonds is found to be greater than \[60{}^\circ \]due to Steric repulsion, between both\[N{{O}_{2}}\], groups, |
| Hence \[{{\mu }_{D}}\] (observed) \[{{\mu }_{D}}\], (Theoretical) |
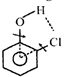 |
| Observed bond angle between C-O and C-Cl bond is found to be less than \[60{}^\circ \] due to intermolecular H-bonding present in it, hence \[{{\mu }_{D}}\] (observed) \[{{\mu }_{D}}\] (Theoretical). |
You need to login to perform this action.
You will be redirected in
3 sec
