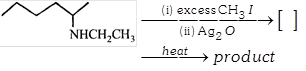
A)
2 and ![]()
B)
2 and ![]()
C) \[~3\text{ }and\,{{H}_{2}}C=C{{H}_{2}}\]
D) \[2and{{H}_{2}}C=C{{H}_{2}}\]
Correct Answer: C
Solution :
[c] The given amine has three \[\beta \]- hydrogen?s, hence it can from three alkenes of which least substituted \[\left( {{H}_{2}}C=C{{H}_{2}} \right)\]will be the major product (Hofmann product or Hotimann elimination). In Hofmann elimination H atom (in the form of proton) is eliminated that \[\beta \]carbon atom which has maximum number of H atom (s). this is due to the fact that Hofmann elimination (an example of E2 reaction) required anti-coplanar arrangement of the \[\beta \]H atom and the leaving group. Further, higher the chances for this arrangement to remain in staggered conformation more will be the ease of elimination. Thus greater the number of H atom at \[\beta \]position, higher will be the chances for \[M{{e}_{3}}{{N}^{+}}-\]and H atom to remain in this particular conformation (anti-coplanar arrangement of \[M{{e}_{3}}{{N}^{+}}-\]and H atom in staggered conformation).You need to login to perform this action.
You will be redirected in
3 sec
