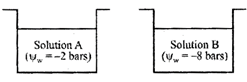
A) Kinetic energy (K.E.) of \[{{H}_{2}}O\] in A solution > K.E. of water in B solution
B) K.E. of water in B solution >K.E. of water in A solution
C) K.E. of water in A solution \[=K.E.\,of\,{{H}_{2}}O\] in b solution
D) Water potential has nothing to do with K.E
Correct Answer: A
Solution :
[a] Water potential (\[{{\psi }_{W}}\]) of solution A is more than that of solution B. Solution having greater water potential possesses more kinetic energy. This kinetic energy is lowered down by addition of solutes.You need to login to perform this action.
You will be redirected in
3 sec
