A) All the carbon in \[{{H}_{2}}C=\underset{\underset{H}{\mathop{|}}\,}{\mathop{C}}\,-C\equiv C-\underset{\underset{H}{\mathop{|}}\,}{\mathop{C}}\,=C{{H}_{2}}\] are in \[s{{p}^{2}}\] hybridisation.
B) In \[{{C}_{2}}{{H}_{2}}{{(CN)}_{2}}\] there are six \['\sigma '\] bonds.
C) In diamond 'C' is in \[s{{p}^{2}}\] hybridisation.
D) In \[{{C}_{3}}{{O}_{2}}\] ail the carbons are in sp hybridisation.
Correct Answer: D
Solution :
(1) 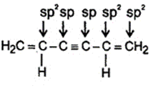 |
| (2) |
| (3) In diamond each carbon atom is in \[s{{p}^{3}}\] hybridisation. |
| (4) \[O=\underset{sp}{\mathop{C}}\,=\underset{sp}{\mathop{C}}\,=\underset{sp}{\mathop{C}}\,=O\] |
You need to login to perform this action.
You will be redirected in
3 sec
