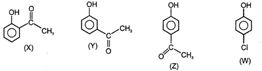
A) \[X>Z>W>Y\]
B) \[Z>X>Y>W\]
C) \[W>Z>X>Y\]
D) \[X>Z>Y>W\]
Correct Answer: B
Solution :
The acidity of phenols increases strongly due to - I, - m group on their ortho and para positions. But para substituted phenols is more acidic than ortho substituted due to intermolecular hydrogen bonding in the ortho substituted phenol.You need to login to perform this action.
You will be redirected in
3 sec
