A)
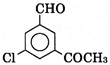
B)
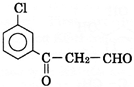
C)
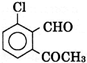
D)
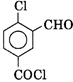
Correct Answer: B
Solution :
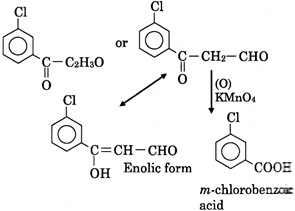
| \[\underset{(A)}{\mathop{{{C}_{9}}{{H}_{7}}{{O}_{2}}Cl}}\,\] exists in keto form and predominatly in enolic form must contain \[-\underset{\underset{O}{\mathop{\parallel }}\,}{\mathop{C}}\,-C{{H}_{{{2}^{-}}}}\]group |
| Oxidation of [A] gives m-chlorobenzoic add |
| \[{{C}_{9}}{{H}_{7}}{{O}_{2}}Cl\,\,(A)\xrightarrow[KMn{{O}_{4}}]{(O)}\] |
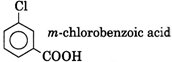 |
| Hence, the compound should have a skeleton struct. |
 |
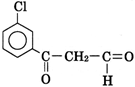
| Thus, A is |
 |
You need to login to perform this action.
You will be redirected in
3 sec
