| Compound \[X({{C}_{3}}{{H}_{6}}O)\] gives negative tests with the following reagents. |
| [a] \[B{{r}_{2}}\] |
| [b] 2, 4-Dinitrophenylhydrazine |
| [c] Na metal. |
| It gives two monochloro structural isomers. Identify 'X'. |
A) \[C{{H}_{2}}=CH-O-C{{H}_{3}}\]
B) \[C{{H}_{3}}-C{{H}_{2}}-CHO\]
C)
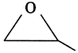
D)
![]()
Correct Answer: D
Solution :
It contains no C=C bond, C=O bond & acidic H.You need to login to perform this action.
You will be redirected in
3 sec
